SEDIA Biosciences Coporation은 감염병 진단과 공중보건 모니터링을 위한 신속 진단키트 및 샘플 수집 기술을 개발하는 미국 바이오 진단 기업입니다.
제품 설명
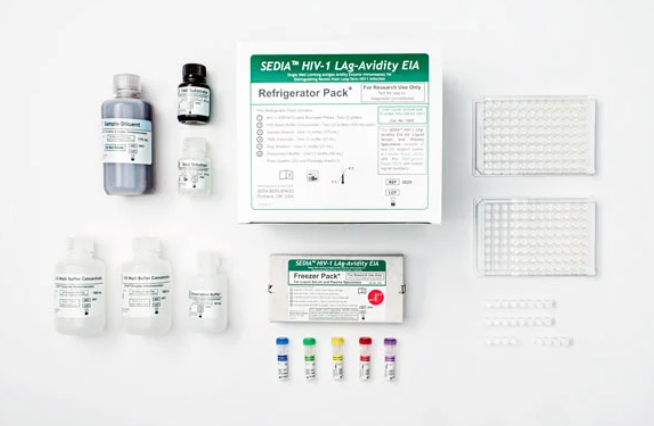
Sedia® HIV-1 Limiting Antigen (LAg)-Avidity EIA
제품 특징
An in vitro single well quantitative limiting antigen avidity enzyme immunoassay for distinguishing recent HIV-1 infections from those which are long-term. Intended for use with liquid serum or plasma specimens.
For Research Use Only: Not for use in diagnostic procedures.
Independent evaluation by CEPHIA showed lowest false recency rate of any HIV incidence assay evaluated.*
Can be used to estimate HIV-1 incidence in a population, monitor and evaluate HIV intervention programs, and recognize those high-incidence populations. Ensures prevention research, vaccine trials, and resources are most appropriately utilized.

| Catalog Number | Description |
|---|---|
| 1002 | Sedia® HIV-1 LAg-Avidity EIA, For Research Use Only |
| Summary of Specifications | |
|---|---|
| Assay Time | 2-4 Hours |
| Processing Temperature | 15° to 37°C (59° to 98.6°F) |
| Storage Temperature | Refrigerator Pack: +2° to 8°C (35.6° to 46.4°F); Freezer Pack: -25° to -10°C (-13° to +14°F) |
| Specimen Type | Human serum or plasma |
| Specimen Volume | 5 µL |
| Custom Antigen | Custom, multi-clade CDC “rIDR-M” HIV-1 gp41 antigen that provides for reduced HIV-1 subtype bias |
| Mean Duration of Recent Infection | Approximately 130 days* |
| False Recency Rate | Less than 1%** |
| U.S. CDC Qualified | Each lot is performance tested by the U.S. Centers for Disease Control prior to market release |
| For Research Use Only | Not for use in diagnostic procedures, RUO products are not to be used for diagnostic purposes, patient management, clinical purposes, or for investigational use within the U.S. |
| HIV Status Requirement | Intended for use only with specimens previously diagnosed as HIV-1 positive |
SEDIA Biosciences Coporation의 모든 제품을 만나 보세요!
Asanté® Rapid Point-of-Care Assays
Asanté® HIV-1/2 Oral Fluid Test
Sedia® HIV Lab Based Products
SEDIA Biosciences Corpoartion - Exclusive Distributor in South Korea "Morebio" 한국 독점 대리점 "모아바이오"